Home
Unit 4
Debye Theory of Specific Heat
Debye Theory of Specific Heat
Introduction
The Debye theory of specific heat was proposed by Peter Debye in 1912
to overcome the limitations of classical theory and Einstein’s model.
It successfully explains the low-temperature behavior of specific heat of solids.
Basic Assumptions
- A solid is treated as a continuous elastic medium.
- Vibrations of atoms are treated as phonons (quantized lattice waves).
- There exists a maximum frequency called Debye frequency.
- Total number of vibrational modes = 3N.
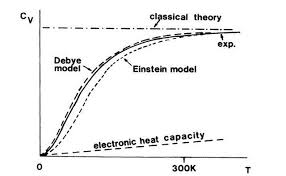
Success of Debye Theory
- Correctly explains low-temperature behavior.
- Predicts \(C_V \propto T^3\).
- Matches experimental results very well.
Important Result
Debye theory successfully explains both:
- Low temperature behavior (\(T^3\) law)
- High temperature classical limit (\(C_V = 3R\))
It provides a complete quantum mechanical description of lattice specific heat.
Derivation of Specific heat as per Debye's Theory :
DEBYES' $T^{3}$ LAW -1912 PETER DEBYE (B.1884-1966, NETHERLAND
As Einstein's Model failed at low temperature region due to his assumption that all the atoms vibrtae with same frequency , Peter Debye came up with idea of Phonons. He considered interaction between the vibrating atoms. Each atom does not vibrate at a constant frquency independently. He suggested that the atoms vibrate cobinedly as coupled oscillators. Their vibration generates a spectrum of frequencies ranging from 0 to a certain value called Debye Frequency ( $f_{D}$ ) . There will be a limit to upper level frequency as all frequecnies are not allowed in the conduction band of the solid. There will be two transverse vibrations and one longitudinal vibrations. The system may be considered as comprising a large Number N of non-inteacting , identical, linear coupled harmonic Oscillators each of mass m , in thermal equillibrium at temperature T.The collective modes of vibrations involving groups of atoms are the possible sound waves which can propagate through solids The energy of vibrational energies of atoms (lattice vibrations ) is quantised and called as phonon. If $f_{i}$ is the frequency of the ith degree of freedom then $E_{n}=h f_{i}\left(n+\frac{1}{2}\right)$
The number of modes of vibration in the frequency range f and $\mathrm{f}+\mathrm{df}$ for three directions of Polarisation ( 2 transverse and 1 longitudinal) .
The number of modes of vibration dN with frequencies in one direction lying in the range f and $\mathrm{f}+\mathrm{df}$ is given by :
$$
\begin{aligned}
E&=\int_{0}^{f_{D}}\langle E\rangle d N\\
&=\int_{0}^{f_{D}} \frac{h f}{e^{\frac{h f}{k T}}-1} \frac{9 n}{f_{D}^{3}} f^{2} d f \\
& =\frac{9 h n}{f_{D}^{3}} \int_{0}^{f_{D}} \frac{f^{3}}{e^{\frac{h f}{k T}}-1} d f\\
&=\frac{9 h n}{f_{D}^{3}}\left(\frac{k T}{h}\right)^{4 x_{m}} \frac{x^{3}}{e^{x}-1} d x\\
&=\frac{9 h n}{f_{D}^{3}} \frac{k T}{h}\left(\frac{k T}{h}\right)^{3 x_{m}} \int_{0} \frac{x^{3}}{e^{x}-1} d x\\
&=\frac{9 R}{f_{D}^{3}} T\left(\frac{k T}{h}\right)^{3 x_{m}} \int_{0} \frac{x^{3}}{e^{x}-1} d x\\
&=\frac{9 R}{f_{D}^{3}} T\left(\frac{k T}{h}\right)^{3 \theta_{D} / T} \int_{0} \frac{x^{3}}{e^{x}-1} d x
\end{aligned}
$$
( where $$x=\frac{h f}{k T}=\frac{k T_{D}}{k T}=\frac{\theta_{D}}{T} $$)
SPECIAL CASES :
Case-1:At high temperature
$$
\begin{aligned}
E &=\frac{9 R}{f_{D}^{3}} T\left(\dfrac{k T}{h}\right)^{3 \theta_{D} / T} \int_{0}^{\dfrac{x^{3}}{x}} d x \\
&=9 R T\left(\frac{k T}{h f_{D}}\right)^{3}\left(\frac{\theta_{D}}{T}\right)^{3} \frac{1}{3} \\
& =9 R T\left(\frac{T}{\theta_{D}}\right)^{3}\left(\frac{\theta_{D}}{T}\right)^{3} \frac{1}{3}=3 R T
\end{aligned}
$$
Thus $$C_{V}=\frac{d E}{d T}=3 R$$
Thus Debye theory reduces to Dulong–Petit law at high temperature.
Case-2: At low temperature
\[
\begin{align}
E&=\frac{9 R}{f_{D}{ }^{3}} T\left(\frac{k T}{h}\right)^{3} \frac{\pi^{4}}{15} \\
& =9 R T\left(\frac{k T}{h f_{D}{ }^{3}}\right)^{3} \frac{\pi^{4}}{15} \\
& =9 R T\left(\frac{T}{T_{D}}\right)^{3} \frac{\pi^{4}}{15}\\
&= \frac{9 R \pi^{4}}{15 \, T_D^3} \, T^4 \\
\Rightarrow C_V &= \frac{dE}{dT} = \frac{36}{15} \, \pi^4 R \left( \frac{T}{\theta_D} \right)^3 \alpha T^3 \\
C_V &\propto T^3
\end{align}
\]
Debye's theory is also known as Debye's $T^{3}$ law. It fits both at high and low temperature. It is observed that at high temperature quantum effects are not of much importance as in each of the three theories specific heat at high temperature is $3 R$.
CRITICISM OF DEBYE'S THEORY:
- The continuum model is valid for long wavelengths (the model assumes only low frequencies are allowed).
- Debye's cut-off frequencies should be different, as longitudinal and transverse waves have different speeds.
- Debye's temperature is assumed to be independent of actual temperature, but it is found to vary up to 10% with temperature.
- Debye's theory assumes that the solid consists of identical atoms, so it is not valid for crystals like NaCl.
- Debye's theory neglects interatomic interaction and electronic contribution to lattice specific heat.
- The assumption of 3N modes of vibration is also not justifiable, as elastic waves are supposed to have infinite frequencies.
- Debye's \( T^3 \) law does not hold well when the temperature is less than 10% of the Debye temperature.

 DMS Learning
DMS Learning

 DMS Learning
DMS Learning